Technologies of CLST
Structural analysis of bio-macromolecular complexes in electron cryo-microscopy
Structural analysis of bio-macromolecular complexes in electron cryo-microscopy (cryo-EM) is a relatively new method in structural biology. In general, the method so-called cryo-EM is the way to examine biological specimens embedded in vitreous ice and particularly, it is aimed to reconstruct three dimensional (3D) structure by processing digital images with computation. There are two major methods in the cryo-EM, one is called cryo-electron tomography and the other is single particle cryo-EM.
Cryo-electron tomography (cryo-ET)
Images observed in transmission electron microscopy are projections of 3D objects. It means that the structural information along Z-axis is overlapped together and sometimes hard to interpret. The method so-called tomography is the way to reconstruct 3D structure from two dimensional (2D) projections to visualize the internal structure. Tomographic reconstruction requires projections from several different angles to compute the 3D structure and the resulting 3D structure can be analyzed as a stack of digital images with certain thickness to clearly visualize the internal structure. The total thickness of the specimen is limited to a few µm with our setup and the thinner specimen results to have higher resolution after the reconstruction.
Here in Protein Structural and Functional Biology Team, we installed a correlative microscope between fluorescence and transmission electron microscopy to utilize fluorescent proteins as fiducial markers to find the area of interest roughly with fluorescent microscope and visualize high resolution structure with electron microscope in the same microscope.
Single particle cryo-EM
Images observed for the purified bio-macromolecular complexes in cryo-EM are assumed as projections of purified, single particle with random orientation. 3D structure of this particle can be reconstructed from certain variety of projections by back-projection method. Recent advances in the detector development and software development give us a chance to obtain near atomic-resolution reconstructions. Then, now the method is of the major interest in the field of structural biology because the single particle cryo-EM can be done without crystallization, with less amount of purified sample, and in close to physiological condition. Recent analysis in high resolution SPR tells that many of specimens in single particle cryo-EM appeared with slightly different conformations, which means that the complexes are flexible in general. For this reason, single particle cryo-EM is now thought to be an additional technique in structural biology, especially for the specimens that are hard to crystallize due to its flexibility.
Here in Protein Structural and Functional Biology Team, we installed dedicated cryo-electron microscope, FEI Tecnai Arctica which is equipped with direct detection device, FEI Falcon2 to accumulate large data set in high quality with automated data acquisition. Then, the high performance computer cluster has been setup to process such a large data set to aim for high resolution structural analysis of bio-macromolecular complexes.
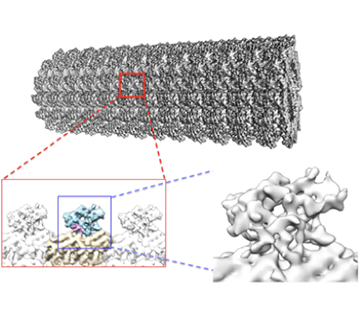
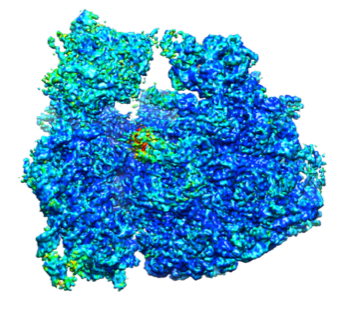
High resolution structure of bacterial ribosome
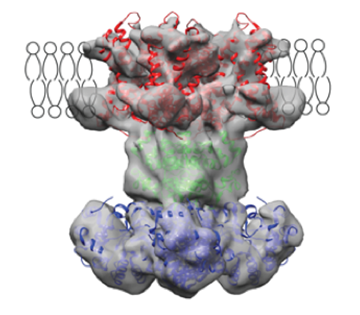
Functional structure of K channel in lipid membrane
High resolution structure of Microtubule & its associated proteins

FEI Tecnai Arctica